


Basic application of polymer in different industries of the world
Polymer has been available since the beginning of human life. Humans used these substances to meet the needs of life without a proper understanding.
These materials (Polymers) are found in nature and all living systems. Among the polymers in the human body are DNA, protein and collagen. Also, polymers found in nature include cellulosic materials such as wood, cotton, linen, wool and so on. Although natural polymers have retained their inherent importance, synthetic polymers are increasingly used today. The first synthetic polymers were synthesized in the second half of the nineteenth century through chemical modification of natural materials, and fully synthetic polymers were developed in the twentieth century. Join us to learn more about these important and practical materials. History of Polymer
In 1833, the Swedish chemist Berzelius coined the wordpolymer for the first time. He referred to the so-called macromolecule after several years of Hermann Staddinger, so he won the Nobel Prize in 1953. In 1930, Wallace Hume Carutz, an American chemist, based Staddinger’s theories on a laboratory basis. In later years, a catalyst was discovered by Karl Ziegler to initiate the polymerization reaction.
Definition of polymers
Polymers, or macromolecules, are large molecules made up of simple duplicate units. The nature and structure of the polymer depends on the monomer or monomers used. If a small number of monomeric units are used to make a polymer, a low molecular weight polymer called an oligomer is produced.
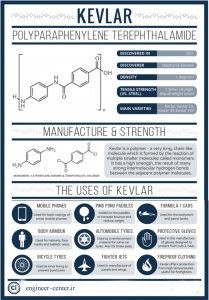
You have probably read about the death of Stephanie Kulk (American chemist) who discovered this polymer
Most of you have probably heard about Kevlar in the field of bulletproof vests. But you know less about its wide range of applications and its robustness.
The discovery of this substance was accidental, Kulk was working for a chemical company (DuPont) and was trying to upgrade a fiber that could be used to make car tires (stronger and lighter).
When he realized that a combination of some of the polymers he was working with made a thin, dark solution
The solution was usually discarded, but he decided to send a sample for testing. He then found that these were much stronger than scalable fibers such as nylon.
However, it was not possible for him to quickly explore Kevlar by developing this fiber.
Kevlar is a polymer (a substance consisting of long chains of materials such as molecules).
Which were usually woven in the form of fibers or cotton fabrics or as a part of composite materials.
Kevlar has a high strength to weight ratio. Hence, it has many uses in human life, including special clothing or ship sails, and so on. If Kevlar weighs as much as steel, it will show 5 times more strength.
KEVLAR fabric can be used as an excellent alternative to carbon fiber or fiberglass. Kevlar composite in light grades, impact resistant, abrasion resistant, heat resistant, and has excellent strength properties for More applications are used.
These polymers are flammable and can be used in the temperature range of about (+90 to -30) degrees Celsius. Due to the high adhesion and tensile strength of hydrogen bonds of amide groups, these polymers are extremely strong.
Also, these polymers with high flexibility have high resistance to petroleum solvents, abrasion, fatigue, high impact and high cracking resistance in sequential bending.
However, due to the polarity of the amide group, they absorb water. The main uses of polyamides in the automotive industry are the production of polymer fibers, sports equipment, electrical circuit boards, the production of textiles and carpets, the manufacture of pipes and hoses, fiber optic cables, and so on.
Polyamides or nylons are engineering plastics that are produced in the following two ways:
1- Comparative polymerization of a diamine and a two-factor acid
2- In the form of polymerization of amino acids
Due to the linear structure of the polyamides, the voids that are present in other random polymers (such as polyethylene) are less present in this polymer, and this factor causes the polymer to have a regular structure and the polymer chains to be located closer to each other.
Of course, these polymers are crystalline on average during injection, but to make fiber and film, the mechanical space between the chains can be reduced by mechanical stretching and the degree of crystallinity can be increased.
Get in touch with us.
Production of General purpose polystyrene with different grades

info@artanpetro.com
Qom Shokouhieh Industrial Town, end of the second phase, Babaei Square, Babaei St., Alam al-Huda St. 1, No. 1331
+982533346396![]()
+982533346473![]()